Your on-line source for reliable and unbiased information about the evaluation & treatment of heart disease.
In 1977 PTCA or balloon angioplasty was introduced as a viable and useful method for treating coronary artery blockages. A deflated balloon was mounted over a skinny catheter and delivered to the coronary artery blockage over a flexible guide wire. The balloon was inflated for about 30 to 60 seconds to open up the blocked passage. However, it suffered from a disadvantage. Although the results looked very good immediately after deflation and removal of the balloon catheter, the blockage would partially return, similar to a sponge becoming larger after being squeezed. This is known as "recoil." Recoil causes the channel to become smaller after having been enlarged by balloon expansion. In other words, a 90% blockage may improve to 20% immediately after PTCA. However, recoil may cause the blockage to worsen and become 30-50% in severity within 15-30 minutes.
Approximately 15 years later, in 1993, coronary stent were introduced to combat "recoil." A stent is a stainless tube with slots. It is mounted on a balloon catheter in a "crimped" or collapsed state. When the balloon is inflated, the stent expands or opens up and pushes itself against the coronary artery blockage. This holds the artery open after the balloon is deflated and removed. Stents behave like a cage that holds the expanded material in place and markedly reduces the amount of recoil.
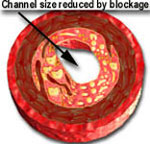
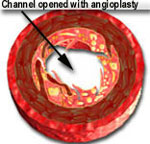
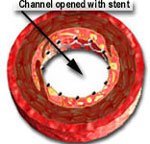
When the balloon is inflated, it expands the stent and opens up the diseased segment into a rounder, bigger and smoother opening (compared to angioplasty, which is shown in the middle picture, above, as having a more "frayed" appearance), Stents induce a more predictable and satisfactory result. It reduces the risk of the artery abruptly closing off during the procedure and also decreases the chance of restenosis (recurrence of the blockage) by nearly 50% (from 30-50% in cases of angiopalsty, down to 15-25% in cases of stents).
Unfortunately, the material within the expanded channel starts to multiply within a few days. This creates a gradual build-up of material that grows through the slots or holes within the stent tube. In 30-60% of cases, the build-up of material can be large enough to cause the blockage to return to its original (or worse) severity in a matter of 6 weeks to 6 months. This known as restenosis.
In April 2003, the U.S. Food and Drug Administration (FDA) approved the first drug-eluting stent to open clogged coronary arteries. . The new stent slowly releases a drug, and has been shown in clinical studies to significantly reduce the rate of re-blockage that occurs with existing stents and angioplasty procedures. The approval was influenced by a U.S. study (called the SIRIUS study), 1058 patients received either the Cypher™ stent or an uncoated conventional stainless steel stent. The Cypher™ Sirolimus-eluting Stent is manufactured and marketed by Cordis pursuant to a license from Wyeth Pharmaceuticals. Sirolimus, the active drug released from the stent, is a naturally occurring substance that reduces the reproduction of tissue that make-up the bulk of restenosis. The drug is cytostatic. Unlike a cytotoxic drug, which kills cells, a cytostatic agent prevents cells from dividing without destroying them, leaving them in a resting, state.
The study demonstrated that after nine months, the patients who received the drug-eluting stent had a significantly lower rate of repeat procedures than patients who received the uncoated stent (4.2% versus 16.8%). Also, patients treated with the drug-eluting stent had a restenosis rate of 8.9%, compared to 36.3% in the patients who received an uncoated stent. The combined occurrence of repeat angioplasty, bypass surgery, heart attacks and death was 8.8% for drug-eluting stent patients and 21% for the uncoated stent patients.
The drug-coated stents are significantly more costly than non-coated stents. However, economic analysis performed independently by researchers such as Dr. David J. Cohen of the Harvard Clinical Research Institute (reported at the American College of Cardiology 52nd Scientific Session in Chicago on March 31, 2003) have confirmed the cost-effectiveness of the CYPHER™ drug-eluting coronary stent. Dr. Cohen used actual hospital in-patient and out-patient cost data, beginning with the period of initial hospitalization and ending one year following stent implantation.
He reported that, "Over the one-year follow-up period, patients who received the CYPHER™ Stent showed substantial reductions in the need for repeat treatments and re hospitalization, translating into substantial post-treatment healthcare savings," said Dr. Cohen. "During 12-month follow-up, for every 100 patients treated with the CYPHER™ Stent, there were 19 fewer revascularization procedures and 25 fewer hospital admissions than with the conventional stent."
In March 2004, Boston Scientific Corporation received approval from the FDA to market its TAXUS™ Express2™ paclitaxel-eluting coronary stent system after demonstrating a significant reduction in the restenosis rate. Other companies, such as Medtronic, are carrying out clinical trials with other types of drug-eluting stents. Insertion of the drug-eluting stent is similar to that of the conventional coronary stent (click on the high-lighted red text to review details of the procedure.
Drug eluting stents and late thrombosis or clot formation: Bare-metal or non-drug coated stents are usually fully coated by body tissue within a few weeks. Once this occurs the stent becomes more resistant to clots and Plavix* can usually be discontinued about 2 months after it is deployed or put in place.
In contrast, drug eluting stents (DES) do not become fully coated with normal tissue for 6 to 12 months, or even longer. Discontinuation of Plavix* prior to this time can result in clot formation and a heart attack. Thus, it is extremely important that patients with a stent (particularly DES) should not stop taking Plavix* until after seeking approval from their cardiologist.
![]()
©1999-2017, 20XXA.S.M. Systems, Inc. All Rights Reserved, including design and all graphic contents & animations
